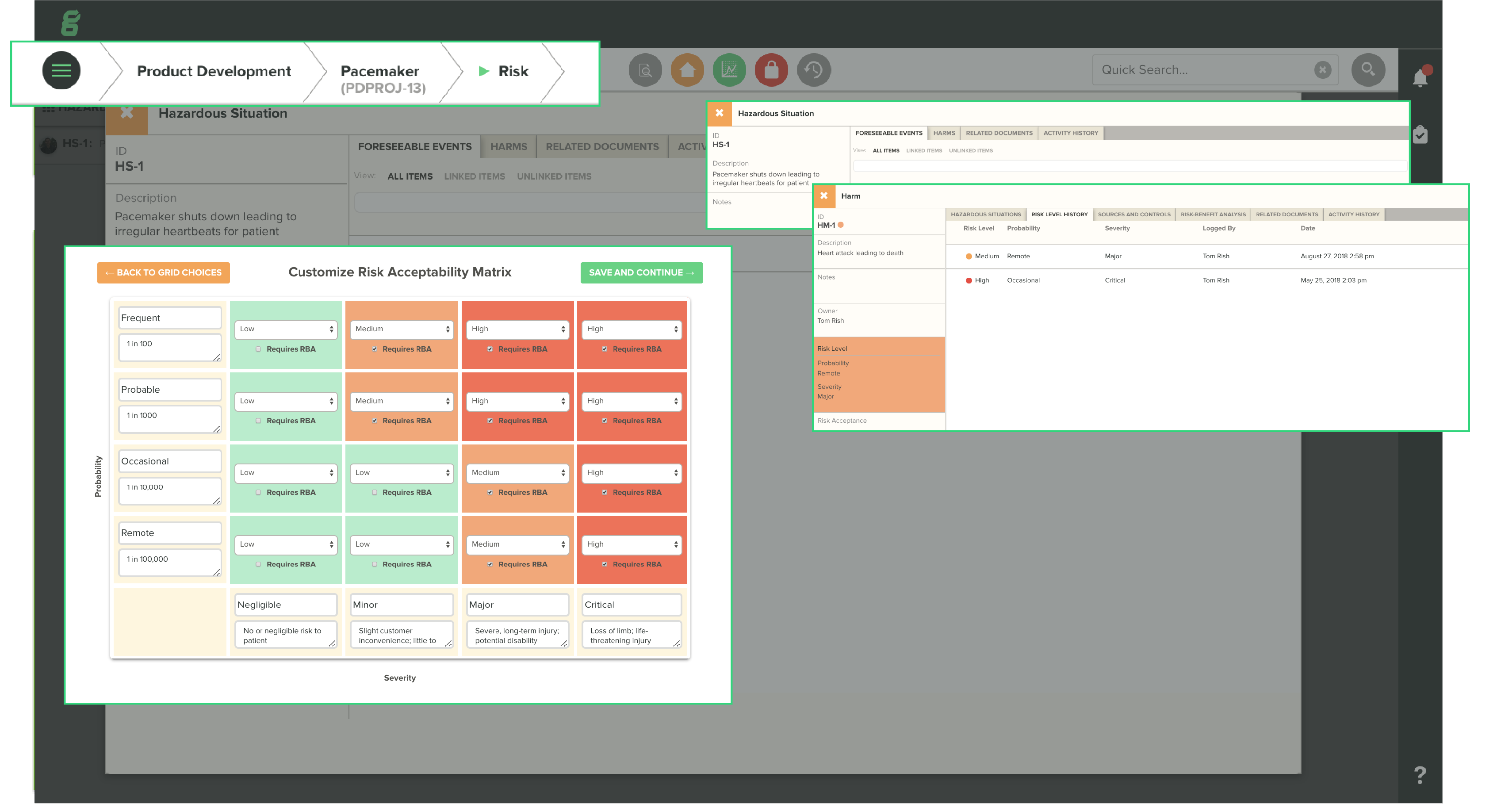
Use Cases and Deployment Scope
We use Greenlight Guru for all of our Quality Management System needs, including design controls, document control, and risk management. Greenlight Guru allows us to develop our medical device products in a way that is compliant with requirements from the FDA and other regulatory bodies and is easily auditable for both internal and external audits.
Alternatives Considered
Qualio and MasterControl Quality Excellence
Other Software Used
Todoist: To-Do List & Task Manager, Asana, Xero